
Digital Pathology Recruitment Agency Europe

Hiring for digital pathology is no longer a “nice to have” capability for scaling healthcare technology firms. In 2026, it is a critical path item that directly affects clinical validation timelines, regulatory submissions, and commercial momentum.
Digital Pathology Recruitment Agency in Europe: Hiring Specialist Talent in 2026
If you are searching for a digital pathology recruitment agency Europe decision-makers can trust, you are usually dealing with one of three situations: you need leadership to de-risk a regulated product roadmap, you need scarce computational pathology talent to move from research to production, or you need cross-border hiring execution across multiple European markets without slowing down.
Digital pathology combines deep clinical context with advanced AI and platform engineering. That hybrid reality makes hiring materially different from generic healthtech, medtech, or software recruitment. The organisations that win in this market treat talent as a strategic system: role design, compensation, assessment, compliance awareness, and a search process built for passive candidates.
What Is Digital Pathology Recruitment?
Digital pathology recruitment is the specialist search and selection of professionals who can build, validate, regulate, and commercialise solutions across the digital pathology stack.
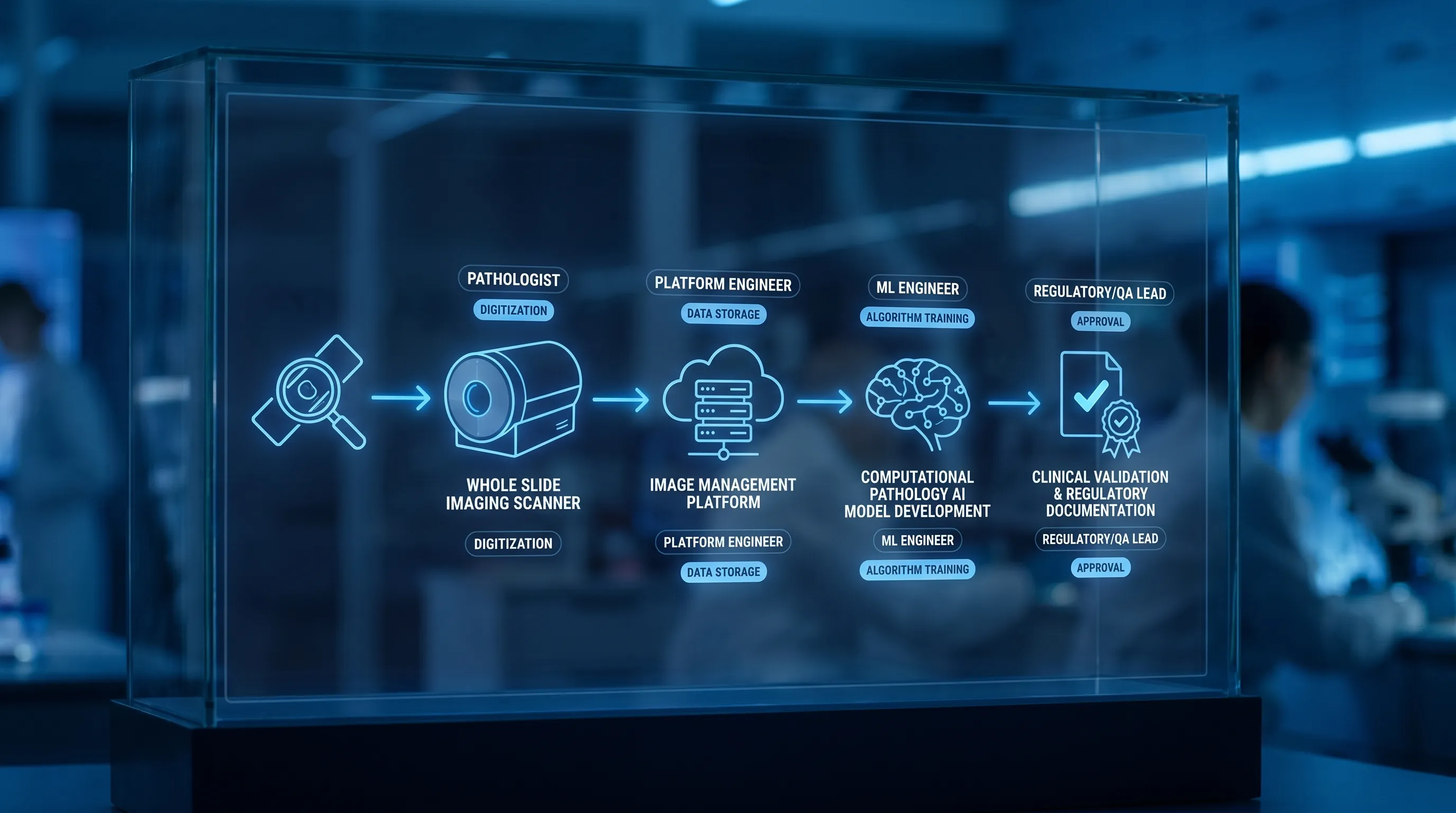
At the technical level, digital pathology typically includes:
- Whole slide imaging (WSI): scanning glass slides into high-resolution digital images.
- Image management and interoperability: storage, retrieval, and workflow integration (often involving DICOM pathology, LIS integrations, and secure data handling).
- Computational pathology: using algorithms to extract quantitative features from tissue images (segmentation, detection, grading support, biomarker quantification).
- AI-powered diagnostics: decision support or diagnostic devices, frequently focused on oncology diagnostics due to data availability and clear clinical value drivers.
Digital pathology recruitment is not the same as hiring for general healthcare IT. A strong engineer in a generic imaging pipeline may not understand clinical evidence standards, dataset governance, or how product decisions impact CE marking. Similarly, a pathologist with research publications may not be ready to operate within an AI product lifecycle, where model monitoring, versioning, and auditability become operational requirements.
Why digital pathology requires a deeply specialist hiring approach
The best digital pathology organisations hire people who can work at the intersection of:
- Clinical workflow reality (lab operations, staining variation, scanner artefacts, inter-observer variability)
- AI development and deployment (data curation, model training, validation strategy, MLOps)
- Regulated product delivery (quality systems, technical documentation, risk management)
That intersection is precisely where the talent shortage is most acute.
Executive search vs contingency recruitment in a digital pathology context
Digital pathology leadership hiring is often a retained, executive search-led process because:
- Many suitable candidates are not actively on the market.
- The role usually carries confidentiality, regulatory, or investor sensitivity.
- The cost of a wrong hire is high (delays to clinical validation, degraded evidence packages, or governance gaps).
Contingency recruitment can work for some mid-level roles when the requirements are narrower and the market is less constrained, but it is typically not optimal for “hybrid-scarce” profiles such as Head of Computational Pathology, VP Engineering in WSI platforms, or VP Regulatory Affairs with AI-device context.
Summary: Digital pathology recruitment is a specialist discipline focused on WSI, computational pathology, and AI-powered diagnostics, where clinical evidence and regulatory constraints shape role design and assessment. It differs from general healthcare technology recruitment because hybrid domain skills are the bottleneck, and executive search is often required for senior and high-risk roles.
Why Hiring Digital Pathology Talent in Europe Is Complex in 2026
Europe is an attractive market for digital pathology companies because it combines world-class academic centres, strong medtech engineering, and large healthcare systems. It is also complex because talent, regulation, and cross-border execution collide.
1) An acute talent shortage in hybrid profiles
Most organisations do not struggle to find “AI engineers” in the abstract. They struggle to find people who can build AI for histopathology under real-world constraints.
The rare profiles tend to be those who can combine:
- Tissue and staining knowledge (H&E staining variations, artefacts, tumour microenvironment concepts)
- Computer vision competence (segmentation, weak supervision, domain generalisation)
- Clinical validation literacy (study design, bias control, ground truth strategy)
- Production readiness (data pipelines, monitoring, model traceability)
In practice, computational pathology recruitment Europe is frequently constrained by a small number of relevant research groups, a limited pool of industry-experienced leaders, and intense competition from adjacent AI medical imaging employers.
2) EU MDR and IVDR pressure increases demand for regulatory-aware hiring
For AI-enabled pathology products that meet the definition of a medical device or in vitro diagnostic, regulatory pathways influence hiring decisions.
- EU MDR (Regulation (EU) 2017/745) raises the bar for clinical evidence, post-market surveillance, and quality system maturity. The regulation text is available via EUR-Lex.
- IVDR (Regulation (EU) 2017/746) similarly increases expectations for evidence, performance evaluation, and lifecycle controls. The regulation text is available via EUR-Lex.
Even when a company has external regulatory consultants, internal hiring still matters. Regulatory work touches product, engineering, clinical affairs, data governance, and documentation. You need leaders who can operationalise compliance, not just interpret it.
3) The EU AI Act changes “good hiring” into “governed hiring”
Digital pathology AI systems are widely treated as high-risk because they are used in a clinical decision context and often connect to regulated device workflows.
The EU AI Act is now part of the operating environment, with phased implementation. Many firms are planning against 2026 milestones to ensure systems, documentation, and governance are audit-ready ahead of enforcement expectations for high-risk use cases. You can reference the Regulation (EU) 2024/1689 text via EUR-Lex.
Hiring implications are direct:
- More demand for risk, validation, and governance capabilities inside ML teams.
- Greater scrutiny on data lineage, bias management, model change control, and technical documentation.
- Increased need for cross-functional leaders who can align engineering, clinical, and quality.
4) Digital pathology is still an emerging sub-sector
Unlike radiology IT or classic LIS vendors, digital pathology has not had decades to produce an established talent pipeline. Many professionals are still transitioning from academia, adjacent medical imaging domains, or enterprise imaging.
That transition is possible, but it requires a search partner who can evaluate transferability rather than rely on simplistic keyword matching.
5) US companies competing remotely for European talent
Even when European companies hire locally, they compete with US firms offering:
- Remote-first senior roles
- Equity-heavy packages
- Global brand credibility in AI
This competition is most visible in senior ML engineering, platform engineering, and product leadership roles.
6) European hubs are competitive and concentrated
In 2026, the market is increasingly hub-driven:
- Cambridge remains a dense cluster due to university pipelines, NHS-linked innovation, and venture activity.
- Paris continues to produce AI-first organisations and research talent.
- Amsterdam remains attractive for international hiring and cross-border teams.
These hubs make sourcing easier, but they also create bidding wars and faster hiring cycles.
Summary: Hiring digital pathology talent in Europe is complex in 2026 because the scarce profiles are hybrid (clinical, AI, regulated delivery), regulatory pressure (EU MDR, IVDR, EU AI Act) increases the cost of mistakes, and competition is global, particularly from US employers hiring European candidates remotely.
Our Strategic Approach to Digital Pathology Recruitment
Optima Search Europe operates as a specialist search partner for business-critical and executive hiring. In digital pathology, the difference between “finding candidates” and “closing hires” is the difference between an organisation that scales on schedule and one that accumulates delivery risk.
Market Mapping & Talent Intelligence
A digital pathology search starts with market mapping, not job advertising.
Market mapping in this niche typically includes:
- Identifying target companies across WSI vendors, AI diagnostics firms, digital health scale-ups, and medtech organisations building pathology modules.
- Mapping research-to-industry transitions (principal investigators, postdocs, lab alumni networks).
- Building a view of “adjacent-fit” pools (computer vision in medical imaging, bioinformatics, regulated software engineering).
This is also where we capture market intelligence that matters to executives:
- Where the talent is concentrated
- Which roles are inflating fastest
- What candidates are rejecting (process length, compensation ambiguity, remote policy, governance maturity)
Executive Search Capability (CTO, VP Engineering, CSO, Head of Computational Pathology)
Digital pathology leadership is often under-hired until late. The cost shows up as:
- Architecture drift in WSI platforms
- Weak clinical evidence strategy
- Late regulatory surprises
- Slow translation from research models to product-grade systems
For leadership roles, we typically run a retained search process designed for passive candidates, including confidential outreach, structured assessment, and close management of stakeholder alignment.
If you are evaluating digital pathology executive search Europe options, prioritise firms that can demonstrate understanding of clinical validation, quality systems, and the technical realities of WSI and computational pathology.
Cross-Border Compliance Expertise
Cross-border recruitment is not just a sourcing exercise. It touches:
- Workforce mobility and relocation constraints
- Right-to-work rules and visa pathways
- Employment models (local entity, EOR, contractor) and misclassification risk
- Data security expectations when hiring remote engineers working with sensitive health data
In regulated healthcare technology, the hiring process itself needs to be designed so that it does not introduce compliance blind spots (for example, unclear responsibilities between ML and quality for documentation and change control).
For organisations building teams across the UK and EU, cross-border execution is a core requirement, not an operational detail.
Salary Benchmarking & Compensation Strategy
Digital pathology compensation is volatile because:
- The candidate pool is thin
- Candidates can move across medtech, AI, and big tech
- Remote offers create cross-border salary anchoring
We advise on compensation structure as well as salary bands. In growth-stage digital pathology firms, the ability to explain equity, progression, and role scope is often what closes the hire.
Candidate Vetting & Assessment Framework
Assessment in this niche must test for real capability, not just CV keywords.
A robust framework typically evaluates:
- Domain understanding: slide preparation variability, artefacts, clinical workflow constraints
- Technical depth: model design choices, segmentation strategy, generalisation, MLOps maturity
- Evidence literacy: validation design, ground truth, performance metrics aligned to clinical claims
- Regulatory awareness: ability to operate under EU MDR/IVDR expectations, and governance thinking aligned with the EU AI Act
- Communication: cross-functional alignment between pathology, engineering, quality, and commercial stakeholders
When appropriate, we recommend work-sample style interviews (short, structured take-homes or live case discussions) that reflect the actual job.
Summary: Our approach combines market mapping, executive search capability, cross-border execution, salary benchmarking, and rigorous candidate assessment. In digital pathology, this integrated method is designed to reduce time-to-hire while mitigating delivery and regulatory risk.
Digital Pathology Roles We Cover
A specialist partner should cover the full organisational spine of digital pathology, not just ML engineers.
In practice, hiring is most successful when you build a balanced team across leadership, engineering, clinical, and regulatory functions.
- Executive & Leadership: CTO, Chief Scientific Officer (CSO), VP Engineering, Head of Computational Pathology, VP Regulatory Affairs
- Engineering & Science: Computational Pathology Scientists, Machine Learning Engineers, Computer Vision Engineers, Bioinformaticians
- Regulatory & Quality: Regulatory Affairs Managers, QA Directors, EU MDR/IVDR Specialists, Data Privacy Officers
- Clinical & Medical: Digital Pathologists, Clinical Affairs Managers, Medical Affairs Directors
- Commercial: VP Sales (Digital Pathology SaaS), Business Development, Market Access, Clinical Affairs (commercial-facing clinical enablement)
Role design matters as much as role coverage. A common failure mode is combining incompatible expectations, for example “Senior ML Engineer” plus “Regulatory documentation owner” plus “Clinical study lead” in one requisition. Those profiles exist, but they are extremely rare and expensive.
Summary: Digital pathology hiring requires coverage across leadership, computational pathology science, ML and platform engineering, regulatory and quality, clinical affairs, and commercial functions. Clear role boundaries and realistic skill combinations materially improve hiring outcomes.
Digital Pathology Recruitment Across Key European Markets
European hiring outcomes vary significantly by market, even when the role title is the same. The differences are driven by talent density, salary expectations, language requirements, and regulatory ecosystem maturity.
United Kingdom (Cambridge and the wider Golden Triangle)
The UK remains a priority market for digital pathology headhunters Europe because it offers high talent density relative to its size.
Cambridge continues to function as a core hub due to:
- Strong university and research pipelines
- Deep health innovation ecosystem
- Proximity to clinical partners and translational research
Companies such as Cyted Health, Histofy, and Spotlight Pathology are often referenced in the ecosystem and contribute to talent circulation through spinouts and leadership moves.
UK hiring considerations in 2026:
- Strong competition for senior ML and platform engineering.
- Candidate caution around long hiring cycles.
- Increased importance of clarity on hybrid working and visa support.
France (Paris)
Paris is producing more AI-first pathology and foundation-model adjacent activity, partly due to the depth of the French AI research community.
The market also benefits from an increasing number of organisations building “platform-first” AI systems, which aligns closely with the needs of computational pathology.
Bioptimus is often discussed in Europe’s AI landscape for a foundation model approach, and more broadly, the Paris ecosystem supports hiring for data, ML, and applied research roles.
France hiring considerations:
- Language requirements vary by function. Engineering is often English-friendly, clinical and regulatory roles are more local.
- Talent quality is high, but process discipline matters because candidates are often evaluating multiple options.
Germany (Heidelberg and Munich)
Germany remains a strong market for regulated medtech hiring due to the depth of engineering talent and a mature compliance culture.
Heidelberg is notable due to academic and clinical research strength, while Munich offers broader engineering and startup density.
Germany hiring considerations:
- Candidates often expect structured organisations (clear role scope, strong leadership, realistic roadmaps).
- Regulatory and quality hiring can be easier than in some other markets, but senior computational pathology leadership remains scarce.
Netherlands (Amsterdam)
Amsterdam continues to be attractive for European headquarters and cross-border teams. It also offers strong English-first hiring conditions and an internationally mobile candidate base.
Netherlands hiring considerations:
- High competition for senior engineers.
- Candidates frequently benchmark offers against US and UK remote roles.
- Relocation and workforce mobility are often easier than in many EU markets, but you still need robust onboarding for international hires.
Belgium (Leuven)
Leuven remains a meaningful cluster due to the academic spinout pipeline from KU Leuven and adjacent research institutions.
Belgium hiring considerations:
- Strong technical and scientific talent.
- Smaller market size means senior hires may be cross-border (Netherlands, France, Germany) or require relocation.
Summary: UK (Cambridge), France (Paris), Germany (Heidelberg/Munich), Netherlands (Amsterdam), and Belgium (Leuven) each offer different advantages for digital pathology hiring. Winning teams build a cross-border strategy rather than relying on a single city, especially for senior computational pathology and regulatory-aware talent.
Digital Pathology Salary Benchmarks in Europe (2026)
Salary benchmarking in digital pathology should be treated as an operating tool, not a static spreadsheet. The market moves with funding cycles, regulatory deadlines, and US remote competition.
The ranges below are indicative and depend on company stage, clinical risk class, evidence maturity, and the candidate’s track record in regulated delivery.
Senior computational pathology scientists vs mid-level ranges
In Europe, computational pathology compensation is driven by scarcity and by whether the candidate has proven industry delivery experience (not just publications).
Indicative base salary ranges (Europe, 2026):
- Mid-level Computational Pathology Scientist: typically in the €70k to €110k base range depending on market
- Senior Computational Pathology Scientist / Staff level: typically in the €110k to €160k base range
Candidates who have led clinical validation strategy, built annotation programmes at scale, or shipped models into regulated workflows can exceed these ranges.
ML and computer vision engineers in digital pathology
ML engineers with WSI experience, large-image pipeline competence, and production MLOps maturity are priced closer to top-tier AI hiring than to classic medtech engineering.
Indicative base salary ranges:
- Mid-level ML/CV Engineer (digital pathology or adjacent medical imaging): €75k to €120k
- Senior ML/CV Engineer (production-grade, strong MLOps, WSI exposure): €120k to €180k
In the UK, senior AI engineers may quote in GBP equivalents that compete with London fintech and big tech. In Amsterdam and Paris, equity and total package structure often matters as much as base.
Regulatory affairs and EU MDR specialist compensation
Regulatory hiring in digital pathology is increasingly specialised because AI introduces additional documentation, change control, and lifecycle complexity.
Indicative base salary ranges:
- Regulatory Affairs Manager (medtech, AI exposure): €80k to €130k
- Senior Regulatory Lead / Head of Regulatory (growth-stage, CE marking ownership): €130k to €200k+
Where IVDR is directly relevant, or where the organisation’s product claims create higher evidence load, packages trend higher.
Geographic differences (UK vs France vs Germany vs Netherlands)
In practical terms:
- UK: high competition, especially Cambridge and London-adjacent markets, with strong upward pressure from US remote offers.
- France: strong research talent, competitive packages for senior AI, sometimes more flexibility in mid-level costs.
- Germany: strong engineering and compliance ecosystem, structured compensation expectations, and careful candidate evaluation of organisational maturity.
- Netherlands: strong international competition, candidates often benchmark against global offers, and total package clarity is critical.
Executive package structures in growth-stage digital pathology companies
Executive compensation is highly context dependent, but usually includes:
- A base salary aligned to local executive norms
- Bonus (often tied to delivery and commercial milestones)
- Equity or long-term incentives (especially Series A to Series C)
For leadership hires, candidates will expect clarity on governance, board expectations, and decision rights. Ambiguity here often kills offers late.
Summary: Digital pathology salary benchmarks in 2026 reflect extreme scarcity in hybrid roles, with senior computational pathology, ML engineering, and regulatory leadership commanding premium packages. Geographic differences matter, but total compensation clarity and credible scope are often the deciding factors in closing senior hires.
Specialized Recruitment Partner vs In-House Hiring
In-house teams can absolutely hire in digital pathology, especially when they have strong employer brand, fast decision-making, and an established candidate pipeline. The question is usually capacity and coverage.
Speed: specialist agency access to passive candidates
A specialist agency’s advantage is not job posting volume. It is the ability to reach passive candidates quickly with credible, informed outreach.
This matters when:
- you need to hire “yesterday” due to regulatory or delivery deadlines
- you are competing with US remote offers
- you need a niche leader where only a small number of people are viable
Market access: candidates not actively on the market
Many strong digital pathology candidates are not applying. They are shipping product, leading research groups, or running teams in adjacent medtech.
A targeted search process (often retained) is designed to engage them respectfully and confidentially.
Risk: wrong hires delay CE marking and clinical validation timelines
In digital pathology, the cost of a wrong hire is not just attrition. It is often:
- clinical study redesign
- model performance surprises late in validation
- documentation gaps discovered during submission preparation
- credibility loss with clinical partners
Compliance awareness per market
Even “good candidates” can be mismatched if they have not operated in regulated environments. A specialist partner screens for regulatory literacy and evidence mindset earlier, reducing downstream surprises.
Executive hiring capacity
Most internal TA functions do not have spare bandwidth for confidential, cross-border C-level search, particularly when the hiring manager is the CTO or the board.
Summary: In-house hiring can work, but specialist digital pathology recruitment is often faster and less risky because it reaches passive candidates, shortens time-to-hire for niche roles, improves assessment quality, and reduces regulatory and delivery risk for high-impact positions.
What Differentiates a Specialized Digital Pathology Recruitment Partner
If you are choosing between a generalist recruiter and a specialist, the decision should be based on measurable capability, not marketing.
Deep sector specialisation
A specialist partner should demonstrate fluency in:
- whole slide imaging workflows
- computational pathology methods (tissue segmentation, detection, grading support)
- the difference between research prototypes and regulated product systems
That fluency improves outreach credibility and reduces false positives.
Executive search capability for regulated and scientific roles
Digital pathology leadership sits across science, engineering, and regulation. A partner must be able to assess executives against that blended success profile.
Multi-country execution and local market knowledge
Cross-border recruitment is where many searches fail. The best partners can execute across the UK and EU markets with realistic expectations on:
- compensation norms
- relocation feasibility
- language requirements
- hiring timelines and notice periods
Real-time market intelligence and salary benchmarking
Market intelligence should show up as:
- guidance on which roles are realistically hireable in 60 days
- compensation strategy that matches the level of scarcity
- advice on role design to avoid impossible requirement stacks
Strategic advisory, not just filling positions
A high-quality partner challenges the brief when needed. For example:
- splitting one “unicorn” role into two realistic hires
- recommending interim leadership while running an executive search
- adjusting interview design to improve signal and reduce drop-off
One practical indicator of a specialist approach is whether the recruiter can discuss your customer landscape credibly. For example, commercial hires may need to understand the buying differences between large hospital systems and private specialist clinics (including international providers such as Laprin Clinic), because sales cycles, procurement, and stakeholder mapping differ materially.
Summary: A specialist digital pathology recruitment partner is defined by sector depth, executive search capability, cross-border execution, live market intelligence, and strategic advisory that improves role design and de-risks hiring decisions.
Case Study / Scenario
The following is a representative scenario based on common hiring patterns in growth-stage digital pathology.
Client profile
- Series B digital pathology company
- Oncology tissue analysis platform
- Based in Cambridge, with expansion hiring across a second European market
Hiring challenge
Hire within 60 days:
- Head of Computational Pathology
- 2 Senior ML Engineers
- Regulatory Affairs Manager
Constraints included an active clinical validation timeline and a CE marking workstream that could not absorb leadership churn.
Process
- European talent mapping across Cambridge, Paris, Amsterdam, and selected German clusters
- Targeted outreach to passive candidates, including adjacent-fit profiles with proven regulated delivery
- Parallel interview tracks to reduce time lost between stages
- Early calibration on compensation, decision rights, and evidence expectations (clinical validation mindset)
Timeline and outcome
- First placement completed in 36 days
- All four roles closed across two markets
- Regulatory and evidence workstreams maintained schedule, avoiding a “stop-start” validation cycle
The key operational lever was not volume. It was speed with accuracy: a shortlist designed around evidence, governance, and real-world product delivery.
Summary: In growth-stage digital pathology, multi-role hiring often fails when companies run serial processes and rely on active candidates. A mapped, cross-border search with parallel interview tracks can close leadership, engineering, and regulatory roles fast enough to protect clinical and CE marking timelines.
Frequently Asked Questions
What does a digital pathology recruitment agency do? A digital pathology recruitment agency sources, evaluates, and closes candidates for roles spanning whole slide imaging platforms, computational pathology science, AI engineering, clinical affairs, and regulatory or quality functions. The key difference versus general recruitment is domain precision: understanding WSI workflows, clinical validation requirements, and the regulated nature of AI-powered diagnostics. A specialist agency also provides market mapping, salary benchmarking, and cross-border hiring execution, which is essential when the best candidates are passive and distributed across multiple European hubs.
How long does it take to hire senior digital pathology talent in Europe? For senior roles, timelines depend on scarcity, interview speed, and compensation clarity. In 2026, a well-run search for a Head of Computational Pathology or a senior ML leader often takes 6 to 12 weeks end-to-end, assuming aligned stakeholders and a decisive process. Delays usually come from unclear success profiles, long gaps between interview stages, or late-stage misalignment on equity and role scope. A specialist search process can reduce time-to-hire by running market mapping and outreach in parallel with interview design.
Which European markets have the strongest digital pathology talent pools? The strongest concentration is typically found around Cambridge (UK), Paris (France), and Amsterdam (Netherlands), with additional depth in German clusters such as Heidelberg and Munich, and a scientific pipeline around Leuven (Belgium). The “best” market depends on role type: senior regulatory and quality leadership is often easier in markets with strong medtech infrastructure, while applied research and ML engineering density can be stronger in AI-heavy hubs. Many companies succeed by hiring cross-border rather than trying to build an entire organisation in one city.
How does EU MDR and the EU AI Act affect digital pathology hiring? EU MDR and IVDR raise expectations for clinical evidence, quality systems, and lifecycle documentation, which increases demand for regulatory-aware engineering leaders, QA professionals, and clinical affairs specialists. The EU AI Act adds governance and risk management requirements that influence how AI teams operate, including documentation, traceability, and validation discipline. In hiring terms, you increasingly need candidates who understand regulated delivery, not just model performance. Organisations also need clearer role ownership across ML, quality, and product to avoid documentation gaps that surface late.
How is digital pathology recruitment different from general healthcare recruitment? Digital pathology recruitment sits at the intersection of pathology workflows, high-resolution imaging platforms, and AI development under regulatory constraints. General healthcare recruitment may not test for WSI-specific technical depth, computational pathology methods, or clinical validation literacy. It may also underestimate the importance of regulatory and evidence experience in technical hires. In digital pathology, a wrong hire can slow clinical studies, disrupt data governance, or create quality system gaps. Specialist hiring focuses on hybrid competence, assessment frameworks that reflect real work, and cross-border sourcing for scarce talent.
Summary: Digital pathology hiring is defined by hybrid skill scarcity, cross-border competition, and increasing governance requirements. A specialist recruitment approach improves speed and reduces risk by combining market mapping, rigorous assessment, and regulatory-aware evaluation.
Conclusion & Strategic Positioning
Digital pathology is moving from early adoption into scaled clinical and commercial deployment. In Europe, that shift is happening under tight constraints: limited hybrid talent supply, global competition for candidates, and regulatory frameworks that demand disciplined delivery.
For CTOs, HR Directors, COOs, founders, and board members, the practical question is not whether you can hire, but whether you can hire fast enough and accurately enough to protect clinical validation, CE marking, and product credibility.
Optima Search Europe supports digital pathology companies with executive search, specialist hiring, and cross-border recruitment execution across European markets, with a focus on business-critical roles where time-to-hire and hiring accuracy materially affect outcomes. If you want to discuss a specific search, role design, or market map, you can learn more at Optima Search Europe.
Summary: In 2026, digital pathology hiring in Europe requires a specialist partner who can combine executive search, computational pathology and AI recruitment depth, cross-border execution, and compliance awareness. The goal is straightforward: reduce time-to-hire while protecting clinical, regulatory, and commercial timelines.

